Problem: Does the amount of baking soda affect the size of the balloon?
Independent variable: Amount of baking soda
Dependent variable: Size of balloon
Research:
Hypothesis: As the amount of baking soda increases, the size of the balloon increases as well.
Materials:
Procedure:
Quantitative Observations:
Data table/graph:
Independent variable: Amount of baking soda
Dependent variable: Size of balloon
Research:
- Baking soda is a chemical reactant
- When mixed with vinegar, gas will be produced.
- Vinegar contains acetic acid, which is reactive.
- The acid mixed with baking soda produces a high amount of gas and bubbles.
Hypothesis: As the amount of baking soda increases, the size of the balloon increases as well.
Materials:
- Baking soda
- Vinegar
- Bottle
- Tablespoon
- Balloons
Procedure:
- Pour 500 ml. of vinegar into bottle.
- Pour 1/4 tablespoons of baking soda into the balloon.
- Attach the balloon top to the bottle top.
- Tip the balloon over until the baking soda falls.
- Observe and record data.
- Clean bottle
- Add 500 ml. of vinegar
- Pour 1/2 tablespoons of baking soda into the balloon
- Repeat steps 3 - 7
- Pour 1 tablespoon of baking soda into the balloon.
- Repeat steps 3 - 7.
Quantitative Observations:
- The bubbles started increasing and gas rose into the balloon
- The smell changed.
- The vinegar started fizzing
- The balloon's size increased accordingly.
Data table/graph:
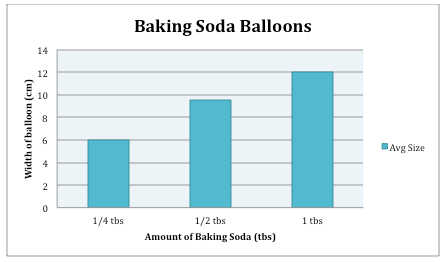
Analysis: As the amount of baking soda is increasing, the bar is getting bigger, showing that the width of the balloon is increasing as well.
Conclusion:
When the amount of baking soda in the vinegar increased, the size of the balloon increased as well. At 1/4 tablespoons, the average width of the balloon was 6 cm. When the amount of baking soda increased to 1/2 tablespoons, the width went up to 9.5 cm. As 1 tablespoon was put in, the width increased to 12 cm. As more baking soda is added, the reaction increases, due to more supply, causing there to be more gas. With more gas, the balloon is blown up bigger. The chemical reaction that occurs between baking soda and vinegar is what causes the gas.
When the amount of baking soda in the vinegar increased, the size of the balloon increased as well. At 1/4 tablespoons, the average width of the balloon was 6 cm. When the amount of baking soda increased to 1/2 tablespoons, the width went up to 9.5 cm. As 1 tablespoon was put in, the width increased to 12 cm. As more baking soda is added, the reaction increases, due to more supply, causing there to be more gas. With more gas, the balloon is blown up bigger. The chemical reaction that occurs between baking soda and vinegar is what causes the gas.
Bibliography citations:
"Chemistry Review: Chemical Change." Chemistry Review. N.p., n.d. Web. 14 Apr. 2013. <http://www.inquiryinaction.org/chemistryreview/chemical_change/>.
Chemistry - A Tale of Vinegar and Hard Water." Everyday Chemistry - A Tale of Vinegar and Hard Water. N.p., n.d. Web. 14 Apr. 2013. <http://www.humantouchofchemistry.com/a-tale-of-vinegar-and-hard-water.htm>
"Chemistry Review: Chemical Change." Chemistry Review. N.p., n.d. Web. 14 Apr. 2013. <http://www.inquiryinaction.org/chemistryreview/chemical_change/>.
Chemistry - A Tale of Vinegar and Hard Water." Everyday Chemistry - A Tale of Vinegar and Hard Water. N.p., n.d. Web. 14 Apr. 2013. <http://www.humantouchofchemistry.com/a-tale-of-vinegar-and-hard-water.htm>